Klinik für Hals-Nasen-Ohren-Heilkunde,
Kopf- und Hals-Chirurgie

Spitzenmedizin und Menschlichkeit im Herzen des Ruhrgebiets
Herzlich willkommen auf der Homepage der HNO-Klinik!
Sehr geehrte Damen und Herren,
herzlich Willkommen auf der Homepage der Universitäts-Hals-Nasen-Ohren-Klinik in Essen. Auf den folgenden Seiten finden Sie Informationen für Patienten sowie Fachinformationen für Ärzte, Wissenswertes über unsere Klinik und ihr Leistungsprofil sowie Möglichkeiten zur schnellen und unkomplizierten Kontaktaufnahme. Weiterhin finden Studierende Aktuelles bezüglich ihrer Kurse und Vorlesungen. Mit 76 Planbetten und knapp 4.000 Eingriffen pro Jahr, sind wir im Einzugsgebiet Essen und darüber hinaus, als Klinik der überregionalen Maximalversorgung, eines der führenden Zentren der deutschen Hochschulmedizin.
Modernste Ausstattung und qualifizierte Mitarbeiter gewährleisten eine gleichbleibend hohe Behandlungsqualität. Durch interdisziplinäre Operationen und Behandlungsstrategien, können hochkomplexe Kopf-Hals-Erkrankungen erfolgreich therapiert werden. Internationale Kooperationsprojekte gewährleisten Spitzenforschung, insbesondere im Bereich der Tumorbehandlung.
Gemäß dem Leitbild des Universitätsklinikums Essen „Spitzenmedizin und Menschlichkeit“ dürfen Sie als Patient neben einer exzellenten medizinischen Versorgung auch menschliche Zuwendung erwarten. Dafür steht das gesamte Team unserer HNO- Klinik.
Mit den besten Grüßen
Univ.-Prof. Dr. med. Stephan Lang




Stellenangebot Audiologie
Als universitäres Klinikum der Maximalversorgung mit einer Kapazität von rund 1.300 Betten betreuen wir mit über 8.000 Beschäftigten in 30 Kliniken, 27 Instituten und Fachzentren jährlich 225.000 Patienten. Unser Haus bietet medizinische Versorgung, modernste Diagnostik und umfassende Therapie mit höchstem internationalem Standard. Hinzu kommt ein umfangreiches Leistungsspektrum in Forschung und…
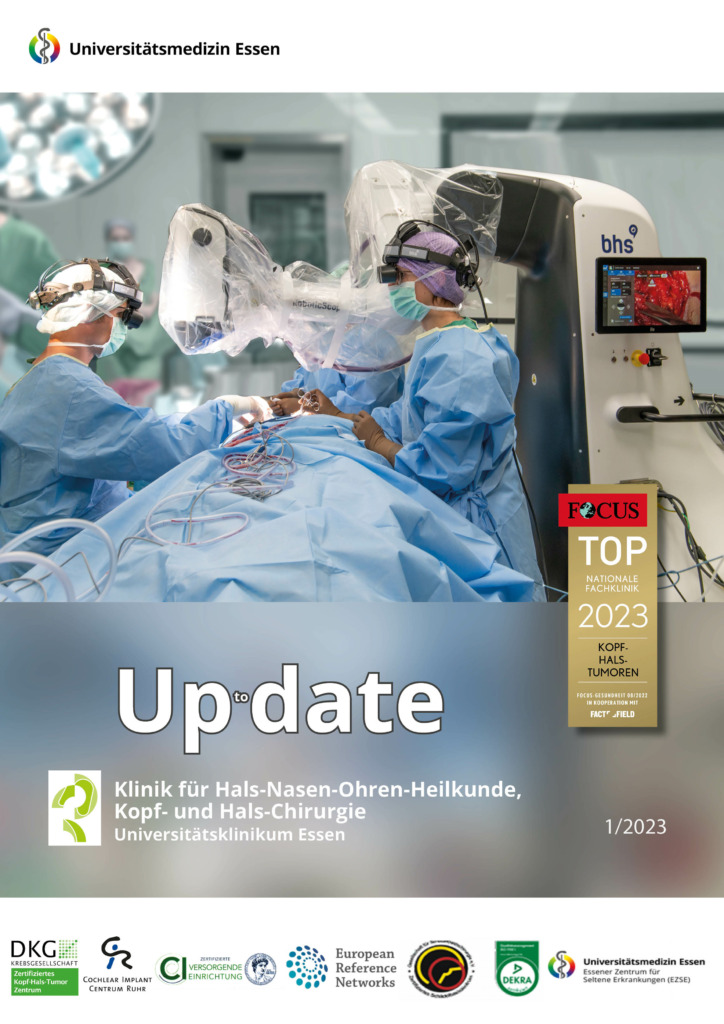
Up to date – unser aktueller Newsletter
Liebe Kolleginnen und Kollegen,es ist mir eine große Freude, Ihnen die aktuelle Auflage unseres Newsletters übersenden zu dürfen. Wie bereits angekündigt, wird die 95. Jahresversammlung der Deutschen Gesellschaft für Hals-Nasen-Ohren-Heilkunde, Kopf- und Halschirurgie, vom 08. bis 11. Mai 2024 in Essen stattfinden. Wir freuen uns sehr, Sie nun offiziell zum…

Zuschlag für Medical Scientist Kolleg: Else Kröner-Fresenius-Stiftung unterstützt Essener Forschungstalente
Die Medizinische Fakultät der Universität Duisburg-Essen (UDE) darf eines von bundesweit nur zwei Medical Scientist Kollegs der Else Kröner-Fresenius-Stiftung (EKFS) aufbauen. Die Forschungsstandorte Essen und Münster setzten sich gegen 28 andere durch und erhalten für vier Jahre jeweils eine Million Euro. Mithilfe der erstmals aufgesetzten Kollegs sollen sich hochtalentierte Naturwissenschaftler:innen…
Rezensionen
Schnelle Aufnahme ohne Termin mit Wartezeit da akute Beschwerden! Woanders muss ich 2 Wochen warten.
Neues Gebäude ganz schick und bequemer Wartebereich. Mit kostenlosen WLAN. Bei der Anmeldung gibts eine Wartenummer, wo man während der Wartezeit am Fernseher verfolgen kann, wenn man dran ist und welcher Raum. Sehr genial. Patientenfragebogen wird digital auf dem Tablet ausgefüllt. Die Arzthelferinnen sind super nett und freundlich. Die Ärztinnen sind auch sehr nett hilfreich und fachlich TOP Beratung und Empfehlungen. Der Hörtest war genial auf der neuesten Technik. Es werrden verschiedene Sounds generiert, um den Tinitus herauszufinden. Schwindeltest mit einer VR Brille fande ich auch super. Mein HNo hat noch ein Geräte aus den 80er Jahren…Sehr zu empfehlen. Einzigster Minuspunkt sind die Parkgebühren auf dem Parkhaus. Das nimmt man aber in Kauf. Für eine gute Behandlung.
Ömer D., Google Reviews, 08.02.2021
Ich war gestern in der HNO Klinik in Essen und war total überrascht, das der Umbau nun fertig war. Hell und freundlich gestaltet, großzügiger Wartebereich, hier war vorher alles sehr gequetscht.
Das Beste ist jedoch die neue Organisation, Anmeldung, Wartemärkchen ziehen, wie beim Straßenverkehrsamt. Alles lief super schnell ab. Zusammen mit Vorgespräch, Hörtest und wieder Arztgespräch war ich in 1,5 Stunden fertig. Wartezeit davor und zwischen den einzelnen Terminen “ 0 „. Kaum hatte ich mich hingesetzt, klingelte die Anzeigetafel mit meiner Nummer schon. Supergründliche Untersuchungen und der Patient ist auf Augenhöhe mit dem Arzt. Ich wurde angehört, ohne Dazwischenreden, um meine Meinung gefragt und ernst genommen. Erste Klasse.
Vor dem Umbau war das alles genauso – nur die Wartezeit war, ohne zu übertreiben, ca. 3 Stunden!!!
Stefanie, Google Reviews, 18.02.2021
Wir sind mehr als zufrieden. Mein Sohn ist dort in der vergangenen Woche 2x wegen einer schweren Gehörgangentzündung behandelt worden. An beiden Tagen waren Ärzte und Schwestern sehr nett und hilfsbereit. Bereits nach der ersten Behandlung kam es zu einer raschen Besserung der Schmerzen. Der niedergelassene HNO hatte das leider nicht hin bekommen.
Steffi Schirsching, Google Reviews, 02/2020